2018 Volume No 36 – pages 81-95
Title: Decellularised tissues obtained by a CO2-philic detergent and supercritical CO2 |
Authors: J Antons, MGM Marascio, P Aeberhard, G Weissenberger, N Hirt-Burri, LA Applegate, PE Bourban, DP Pioletti |
Address: EPFL/STI/IBI/LBO, Station 9, 1015 Lausanne, Switzerland |
E-mail: dominique.pioletti at epfl.ch
|
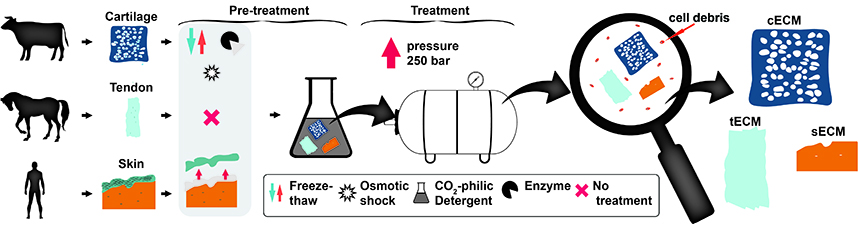
Abstract: Tissue decellularisation has gained much attention in regenerative medicine as an alternative to synthetic materials. In decellularised tissues, biological cues can be maintained and provide cellular environments still unmet by synthetic materials. Supercritical CO2 (scCO2) has recently emerged as a promising alternative decellularisation technique to aggressive detergents; in addition, scCO2 provides innate sterilisation. However, to date, decellularisation with scCO2 is limited to only a few tissue types with low cellular density.
|
Key Words: Extracellular matrix, cell sheet, osteoinductive capacity, osteoconductive capacity, bone regeneration, non-union model. |
Publication date: September 4th 2018 |
Article download: Pages 81-95 (PDF file) |