1 Tissue Engineering Research Group (TERG), Department of Anatomy and Regenerative Medicine, Royal College of Surgeons in Ireland (RCSI), D02 YN77 Dublin, Ireland
2 Trinity Centre for Biomedical Engineering (TCBE), Trinity College Dublin, D02 R590 Dublin, Ireland
3 Advanced Materials and BioEngineering Research (AMBER) Centre, D02 YN77 Dublin, Ireland
4 Regenerative Medicine & Cellular Therapies Division, The University of Nottingham Biodiscovery Institute (BDI), School of Pharmacy, University of Nottingham, NG7 2RD Nottingham, UK
5 NIHR Nottingham Biomedical Research Centre, University of Nottingham, NG7 2RD Nottingham, UK
Abstract
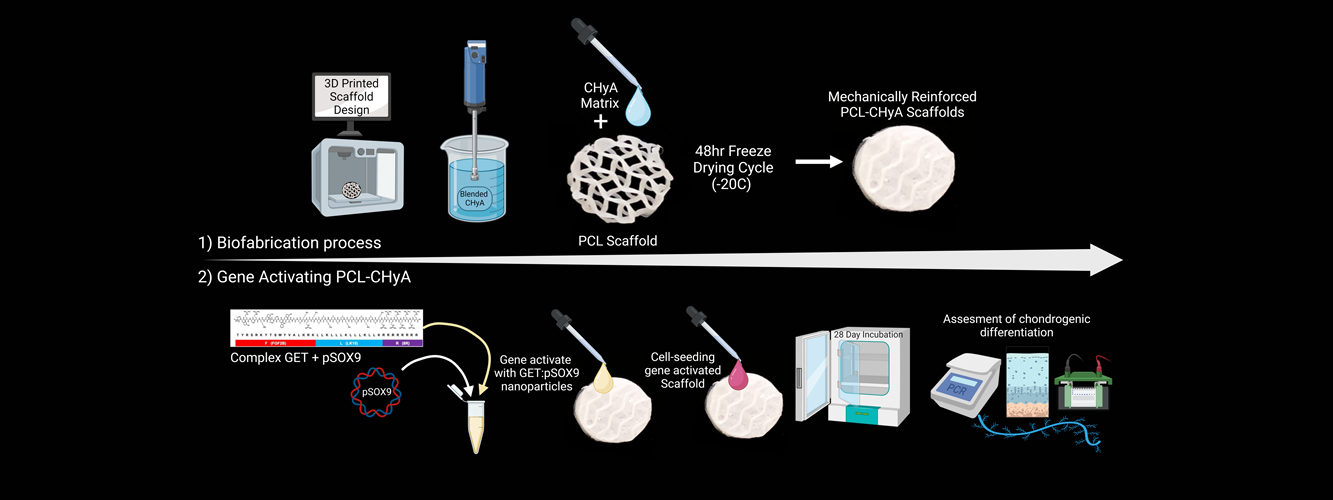
Articular cartilage (AC) has a poor capacity to repair once damaged, with progressive degeneration often leading to osteoarthritis (OA). While biomaterials fabricated with extra cellular matrix (ECM) native to the AC have shown promise for repair of focal AC defects, several challenges must be overcome for the repair of larger load bearing defects due to poor scaffold mechanical properties and a lack of chondrogenic potential in diseased cells. Here, we develop a method to improve such biomaterials by incorporating a bioabsorbable 3D printed reinforcing framework and the delivery of pro-chondrogenic genes to infiltrating stem cells to enhance chondrogenesis and produce hyaline tissue that is more indicative of healthy AC. A bioabsorbable polycaprolactone (PCL) 3D printed framework was surface treated to improve its hydrophilicity and used to reinforce a collagen hyaluronic acid (CHyA) matrix. The mechanically reinforced scaffolds were then gene-activated (GA) with the chondrogenic transcription factor SOX9 which was complexed with non-viral nanoparticles (NPs) generated using the glycosaminoglycan-binding enhanced transduction (GET) system, before being seeded with human mesenchymal stromal cells (hMSC). After 28 days culture in chondrogenic media, hMSCs on the GA-scaffolds deposited an ECM more indicative of healthy hyaline cartilage compared to the gene free control. SOX9 mRNA expression on the GA scaffold was 2-orders of magnitude higher than on the control, with downstream chondrogenic targets of SOX9 (COL2α1, ACAN) also expressing significantly higher mRNA levels. Expression of pro-chondrogenic ECM proteins such as COL2, were 17.5 times greater (p = 0.0018) on the GA scaffold which also resulted in enhanced production and spatial distribution of sulphated glycosaminoglycans (sGAG), which are critical to the function of healthy AC. In summary, these findings provide evidence that functionalization of a 3D printed biomimetic pro-chondrogenic scaffold with SOX9 NPs enhances the quality of ECM produced by human stem cells on such mechanically reinforced scaffolds.
Graphical Abstract
Keywords
- SOX9
- gene activated scaffold
- non-viral vector
- pro-chondrogenic
- bioregenerative
- cartilage repair
