1 Department of Rehabilitation Medicine, The Second Affiliated Hospital of Anhui Medical University, 230601 Hefei, Anhui, China
2 Department of Rehabilitation Medicine, The Second Affiliated Hospital of Wannan Medical College, 241000 Wuhu, Anhui, China
3 Research Center for Translational Medicine, The Second Affiliated Hospital of Anhui Medical University, 230601 Hefei, Anhui, China
Abstract
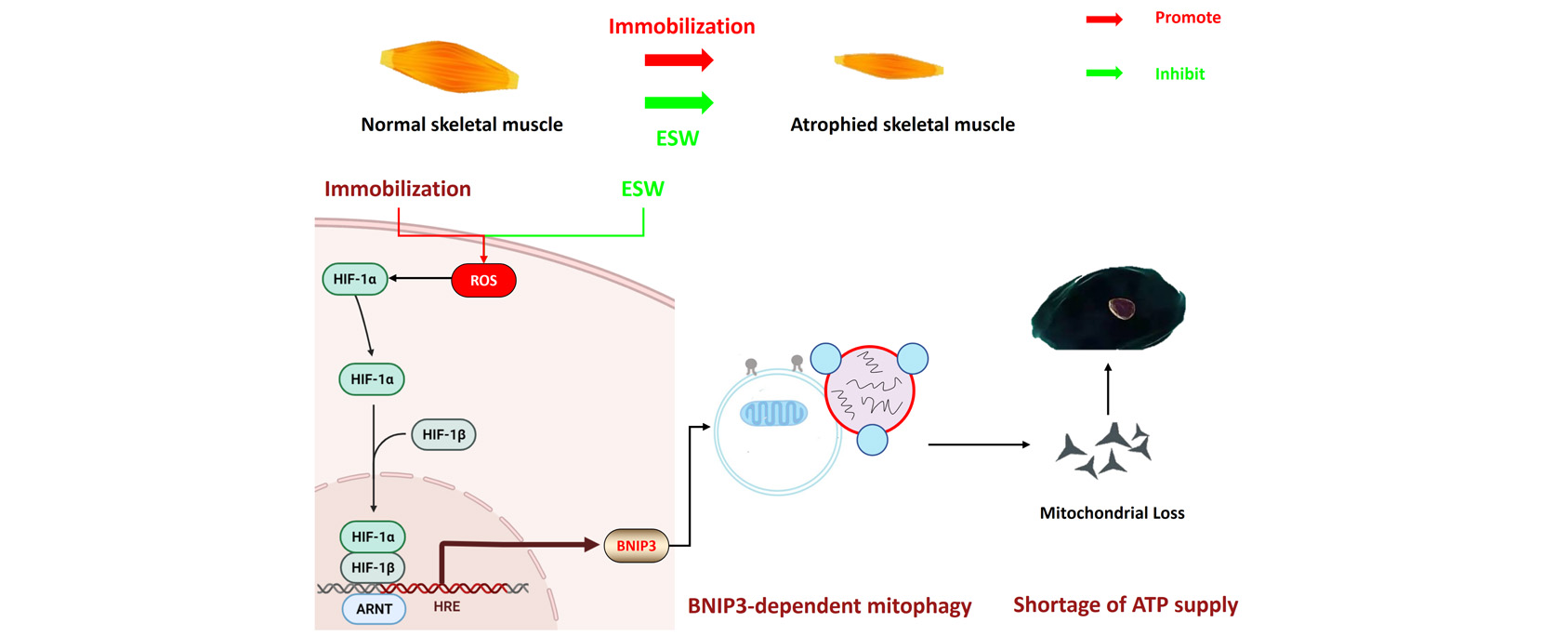
Purpose: The purpose of this study is to investigate the role of Bcl2/adenovirus E1B 19kDa protein-interacting protein 3 (BNIP3)-dependent mitophagy in immobilization-induced muscle atrophy and explore the potential mechanism responsible for ameliorating myogenic contracture through extracorporeal shock wave (ESW). Methods: A rat model of myogenic contracture was developed by immobilizing the knee joint, and muscle atrophy and mitophagy were assessed in vivo . Subsequently, we verified the role of mitophagy in muscle atrophy through in vitro . Finally, we examined the potential therapeutic effects of ESW on immobilization-induced muscle atrophy and myogenic contracture, and assessed the role of mitophagy in the observed alleviation of symptoms. Results: Immobilization had significant time-dependent effects on muscle atrophy. It triggered the production of excessive reactive oxygen species (ROS) and subsequently overactivated hypoxia-inducible factor (HIF)-1α/BNIP3-dependent mitophagy in the rectus femoris. Excessive mitophagy resulted in the elimination of mitochondria, leading to a shortage of adenosine triphosphate (ATP). As ATP homeostasis was crucial for maintaining muscle mass, the shortage of ATP resulted in reduced muscle mass and cross-sectional area, as well as slow-to-fast myofiber type transition. These findings were corroborated by an in vitro study that pretreatment with BNIP3 small interfering RNA (siRNA) reduced the level of BNIP3-dependent mitophagy and partially counteracted the inhibition of myogenic differentiation caused by high levels of ROS. Notably, ESW was found to suppress ROS generation, inhibit overactivated HIF-1α/BNIP3-dependent mitophagy, and preserve mitochondrial quantity in the rectus femoris. Conclusions: Our findings suggest that ESW can inhibit overactivated BNIP3-dependent mitophagy in immobilized muscles, which could be one of the potential therapeutic mechanisms attenuating immobilization-induced muscle atrophy and further improving myogenic contracture.
Graphical Abstract
Keywords
- Muscle atrophy
- myogenic contracture
- immobilization
- mitophagy
- extracorporeal shock wave
