Biomimetic neuropeptide Y/collagen I/β-tricalcium phosphate scaffold mediated macrophage polarization and vascularization for bone regeneration
1 Department of Orthopedics, The 6th Affiliated Hospital of Shenzhen University Health Science Center, 518052 Shenzhen, Guangdong, China
2 Department of Orthopedics, The 8th Affiliated Hospital of Sun Yat-Sen University, 518000 Shenzhen, Guangdong, China
3 Department of Pain, The 8th Affiliated Hospital of Sun Yat-Sen University, 518000 Shenzhen, Guangdong, China
§These authors contributed equally.
Abstract
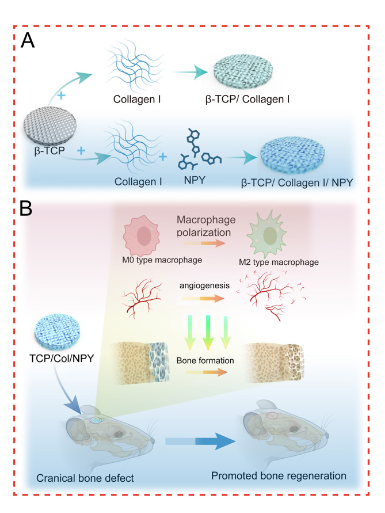
Background: Bone grafting is the primary clinical intervention for bone defects. β-tricalcium phosphate (β-TCP) is an absorbable ceramic for its excellent biocompatibility and bioactivity. Neuropeptide Y (NPY) participates in bone homeostasis and vascular regeneration. This research explored the TCP/collagen I (Col)/NPY scaffolds, which controlled release of NPY, on macrophage polarization during bone repair. Methods: The scaffold was characterized by scanning electron microscopy (SEM) and Fourier-transform infrared (FTIR) spectrometry. The cumulative NPY release from the TCP/Col/NPY scaffold was tested by an enzyme-linked immunosorbent assay. Biocompatibility of the TCP/Col/NPY scaffold was evaluated using cell counting kit (CCK)-8 and calcein-acetoxymethyl ester (AM)/propidium iodide (PI). Angiogenic activity was detected by scratch and tube-formation assays with human umbilical vein endothelial cells (HUVECs). Osteogenic differentiation was detected by alkaline phosphatase (ALP) staining. Flow cytometry and immunofluorescence staining were used to evaluate RAW264.7 polarization. In vivo, bone-defect repair was evaluated using micro-computed tomography scans, hematoxylin and eosin (H&E) staining, Masson and immunohistochemical staining. Results: The SEM images disclosed an interconnected pore structure. FTIR of TCP/Col/NPY scaffolds showed a characteristic peak of NPY. The TCP/Col/NPY scaffold exhibited favorable biocompatibility with bone marrow mesenchymal stem cells and promoted the migration and angioenesis of HUVECs. RAW264.7 upregulated cluster of differentiation-206 (CD206) in the TCP/Col/NPY group (p < 0.05). In vivo, the TCP/Col/NPY scaffold promoted the repair of cranial defects. H&E and Masson revealed that new bone formation in TCP/Col/NPY group was significantly higher (p < 0.05). Runx2, osteocalcin, platelet-derived growth factor-BB and CD206/CD80 expression were higher in the TCP/Col/NPY group (p < 0.05). Conclusions: TCP/Col/NPY scaffolds significantly promoted the repair of critical bone defects by modulating macrophage polarization toward the M2 phenotype and enhancing vascular regeneration.
Graphical Abstract
Keywords
- Tricalcium phosphate
- neuropeptide Y
- bone repair
- M2-type macrophages
