Photo-crosslinked hydrogel for enhanced bone defect regeneration via sustained release OGP(10-14)
1 Department of Biomedical Materials Science, School of Biomedical Engineering, Third Military Medical University, 400038 Chongqing, China
2 Guangdong Key Laboratory for Biomedical Measurements and Ultrasound Imaging, National-Regional Key Technology Engineering Laboratory for Medical Ultrasound, School of Biomedical Engineering, Medical School, Shenzhen University, 518060 Shenzhen, Guangdong, China
3 State Key Laboratory of Trauma and Chemical Poisoning, Third Military Medical University, 400038 Chongqing, China
§These authors contributed equally.
Abstract
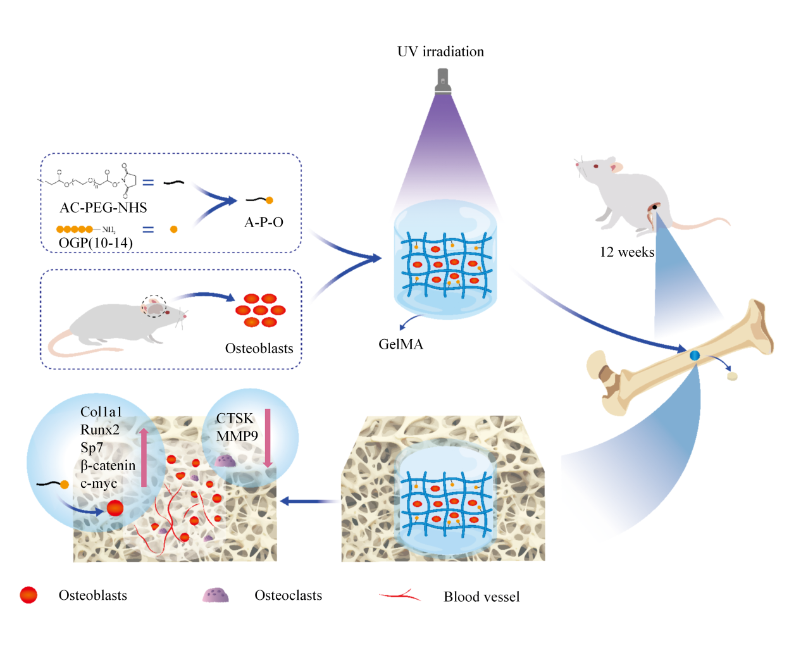
Background: Currently, the clinical treatment of severe bone defects remains a major challenge. Gelatin methacryloyl (GelMA) hydrogels with extracellular matrix (ECM)-like properties are commonly used materials for bone defect repair. However, due to the lack of osteogenic activity, researchers have considered using tissue engineering methods to address this issue. Methods: A composite hydrogel scaffold was fabricated by incorporating acryloylated polyethylene glycol N-hydroxysuccinimide (AC-PEG-NHS)-modified osteogenic growth peptide (OGP)(10-14) (AC-PEG-OGP(10-14)) and primary osteoblasts into a GelMA matrix. The hydrogel scaffold was characterized using scanning electron microscopy (SEM), mechanical testing, and the bicinchoninic acid (BCA) protein assay. In vitro, the effects of the scaffold on the differentiation of osteoblasts and bone marrow-derived macrophages (BMMs) were evaluated. In vivo, its role in bone defect repair was assessed using a rat model. Results: The results demonstrated that the AC-PEG-OGP(10-14)-loaded hydrogel scaffold significantly enhanced the mechanical properties and slowed the degradation rate based on the GelMA scaffold, and it could also achieve the controlled release of the osteogenic peptide OGP(10-14). In vitro and in vivo experiments showed that the material exhibited excellent biocompatibility and osteogenic mineralization properties. It inhibited osteoclast formation, thereby significantly promoting the repair of mouse femoral bone defects. The underlying mechanism is closely related to the activation of the Wnt/β-catenin signaling pathway. Conclusions: AC-PEG-OGP(10-14)-loaded hydrogel scaffold offers an effective solution for repairing bone defects and significantly enhances bone regeneration.
Graphical Abstract
Keywords
- GelMA
- OGP(10-14)
- bone defect repair
- tissue engineering
