1 Department of Gynecology, Cangnan Affiliated Hospital of Wenzhou Medical University, 325800 Wenzhou, Zhejiang, China
2 Department of Radiation Oncology, Wenzhou Geriatric Hospital, 325000 Wenzhou, Zhejiang, China
3 Department of Radiation Oncology, The First Affiliated Hospital of Wenzhou Medical University, 325000 Wenzhou, Zhejiang, China
4 Zhejiang South Institute of Radiation Medicine and Nuclear Technology Application, 325809 Wenzhou, Zhejiang, China
Abstract
Background: Endometrial cancer (EC) is a prevalent gynecological malignancy with limited therapeutic options due to drug resistance and systemic toxicity. Exosomes have emerged as promising targeted drug delivery vehicles, but their application in EC remains underexplored. Thrombospondin-1 (THBS1), a key regulator of extracellular matrix remodeling and transforming growth factor β (TGF-β) signaling, may offer a novel therapeutic strategy for EC. Methods: Differentially expressed genes were identified from the public transcriptomic datasets and subjected to GO and KEGG enrichment analyses. Exosomes derived from Human embryonic kidney 293 (HEK293) cells overexpressing THBS1 were isolated and characterized by Western blotting, transmission electron microscopy, and nanoparticle tracking analysis. Their effects on EC cells were assessed using Cell Counting Kit-8, 5-ethynyl-2’-deoxyuridine incorporation, colony formation, wound healing, and cell invasion assays. TGF-β pathway involvement was assessed using the agonist SRI-011381. In vivo experiments were conducted to evaluate tumor growth, epithelial–mesenchymal transition (EMT) marker expression, and treatment safety. Results: THBS1 expression was downregulated in the EC cells. The restoration of THBS1 expression through exosome delivery inhibited cell proliferation, migration, and invasion. THBS1-overexpressing exosomes attenuated the activation of the TGF-β/Smad pathway and modulated EMT-related markers. The inhibitory effects of THBS1-exosomes were reversed by SRI-011381. In vivo, THBS1-exosome treatment markedly suppressed tumor growth, enhanced E-cadherin expression, reduced vimentin levels, and exhibited no observable toxicity. Conclusions: Exosomes overexpressing THBS1 effectively suppressed EC progression by targeting the TGF-β/Smad signaling axis and EMT. These findings support the potential of THBS1-enriched exosomes as a novel, safe, and targeted therapeutic modality for future EC.
Graphical Abstract
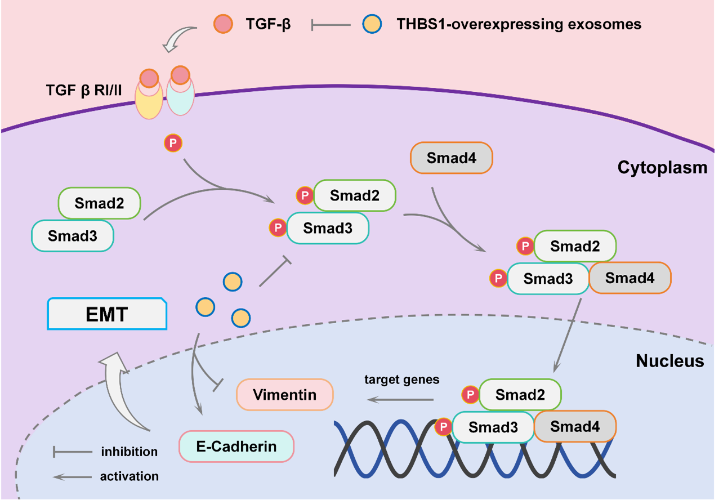
Keywords
- Endometrial cancer,
- thrombospondin-1
- exosomes
- TGF-β/Smad signaling
- epithelial-mesenchymal transition
