Pulsed electromagnetic fields synergize with graphene to enhance dental pulp stem cell-derived neurogenesis by selectively targeting TRPC1 channels
1 Faculty of Dentistry, National University of Singapore, Singapore
2 Molecular Neuropsychiatry & Development (MiND) Lab, Campbell Family Mental Health Research Institute, Centre for Addiction and Mental Health, Toronto, ON, Canada
3 Institute of Medical Science, University of Toronto, Toronto, ON, Canada
4 Department of Surgery, Yong Loo Lin School of Medicine, National University of Singapore, Singapore
5 Biolonic Currents Electromagnetic Pulsing Systems Laboratory, BICEPS, National University of Singapore, Singapore
6 Department of Pharmacy, National University of Singapore, Singapore
7 Craniofacial Research and Innovation Center, National University of Singapore, Singapore
8 Department of Physiology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore
9 Institute for Health Innovation & Technology, iHealthtech, National University of Singapore, Singapore
10 Healthy Longevity Translational Research Programme, Yong Loo Lin School of Medicine, NUS, Singapore
§ These authors equally contributed to this paper
Abstract
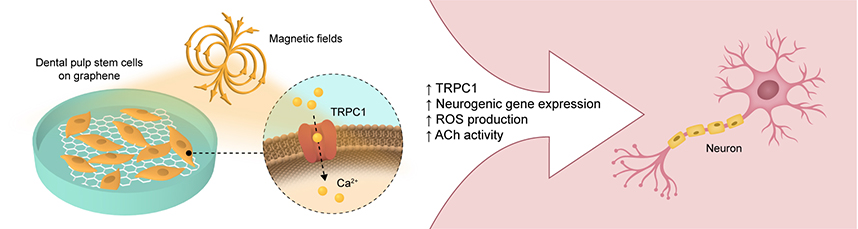
Conventional root canal treatment replaces the infected pulp with defined materials. Alternative cell-based tissue engineering strategies aim to regenerate a fully functional pulp within the root canal. Despite recent advances in this area, however, the regeneration of an innervated pulp remains a major challenge in the field. Both graphene (2DG) and pulsed electromagnetic fields (PEMFs) independently have been shown to promote diverse cellular developmental programs. The present study showed that 2DG promoted the neurogenic induction of human dental pulp stem cells (hDPSCs) by upregulating and accelerating the expression of mature neuronal markers. Notably, 2DG induced the highest expression of transient receptor potential canonical cation channel type 1 (TRPC1) during early neurogenesis. As brief PEMF exposure promotes in vitro differentiation by activating a TRPC1-mitochondrial axis, an opportunity to combine 2DG with developmentally targeted PEMF exposure for synergistic effects was realizable. Neurogenic gene expression, neurotransmitter release, and reactive oxygen species (ROS) production were greatly enhanced by a brief (10 min) and low amplitude (2 mT) PEMF exposure timed to coincide with the highest TRPC1 expression from hDPSCs on 2DG. In contrast, hDPSCs on glass were less responsive to PEMF exposure. The capacity of PEMFs to promote neurogenesis was precluded by the administration of penicillin/streptomycin, mirroring previous studies demonstrating that aminoglycoside antibiotics block TRPC1-mediated calcium entry and verifying the contribution of TRPC1 in this form of magnetoreception. Hence, graphene created a more conducive environment for subsequent PEMF-stimulated neurogenic induction of hDPSCs through their mutual capacity to activate TRPC1with subsequent ROS production.
Graphical Abstract
Keywords
- Pulsed electromagnetic fields
- mitohormesis
- tissue engineering
- nanomaterial
- pulp regeneration
