1 Trinity Centre for Biomedical Engineering, Trinity Biomedical Sciences Institute, Trinity College Dublin, D02 R590 Dublin, Ireland
2 Department of Mechanical, Manufacturing and Biomedical Engineering, School of Engineering, Trinity College Dublin, D02 PN40 Dublin, Ireland
3 Advanced Materials and Bioengineering Research Centre (AMBER), Royal College of Surgeons in Ireland and Trinity College Dublin, D02 W9K7 Dublin, Ireland
4 Altach Biomedical Limited, D04 C5Y6 Dublin, Ireland
5 School of Biochemistry and Immunology, School of Medicine, Trinity Biomedical Sciences Institute, Trinity College Dublin, D02 R590 Dublin, Ireland
6 School of Mechanical and Materials Engineering, Engineering and Materials Science Centre, University College Dublin, D04 V1W8 Dublin, Ireland
7 UCD Centre for Biomedical Engineering, University College Dublin, D04 V1W8 Dublin, Ireland
8 Conway Institute of Biomolecular and Biomedical Research, University College Dublin, D04 V1W8 Dublin, Ireland
9 Orthopaedics and Sports Medicine, School of Medicine, Trinity College Dublin, D02 PN40 Dublin, Ireland
10 Sports Surgery Clinic, D09 C523 Dublin, Ireland
11 School of Veterinary Medicine, University College Dublin, D04 V1W8 Dublin, Ireland
Abstract
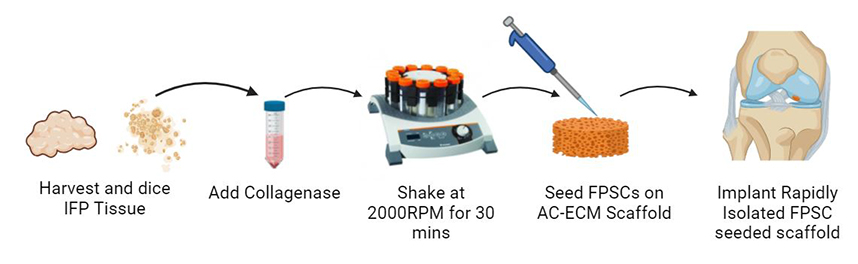
Background: Clinically approved cell-based cartilage repair products are associated with multiple surgeries and high cost. There is therefore an unmet clinical need for a cell-based approach that is efficacious and cost effective that can be performed intraoperatively as a single-stage procedure. Methods: Here, we developed a novel methodology to rapidly isolate chondro-progenitor cells from human infrapatellar fat pad tissue that reduced the isolation time from over three hours to under one hour while still being able to yield clinically relevant cell numbers. Cell yields and biochemical characteristics were compared with conventionally isolated control cells. In vitro assays evaluated cartilage-specific matrix deposition across multiple human donors. Constructs combining rapidly isolated cells and articular cartilage extracellular matrix-derived scaffolds were implanted into caprine cartilage defects and analyzed after six months in vivo . Results: These rapidly isolated cells contained a larger fraction of colony-forming cells than conventionally isolated control cells, and an analysis of surface marker expression revealed a higher percentage of CD44 + (a putative progenitor cell marker) cells in this group. Furthermore, these rapidly isolated cells supported higher levels of cartilage-specific matrix deposition in vitro for multiple human donors. We then seeded such rapidly isolated cells onto cartilage extracellular matrix (ECM) derived scaffolds and immediately (i.e., no in vitro pre-culture) implanted these constructs into caprine cartilage defects. After 6 months in vivo , treatment with this cell and scaffold combination typically generated a repair tissue that was rich in glycosaminoglycans and type II collagen, with biomimetic collagen fiber alignment and lubricin expression in the superficial zone, which was generally not observed in defects treated with microfracture. However, in this model the addition of the rapidly isolated cells did not result in any significant improvement in repair metrics compared to treatment with the extracellular matrix scaffold alone. Conclusions: While this rapidly isolated cell population processes a strong chondrogenic potential in vitro , further work is required to identify clinical scenarios where it will provide clear therapeutic benefits.
Graphical Abstract
Keywords
- Cartilage repair
- articular cartilage
- stem cell therapy
- tissue engineering
- regenerative medicine
- biomaterials
